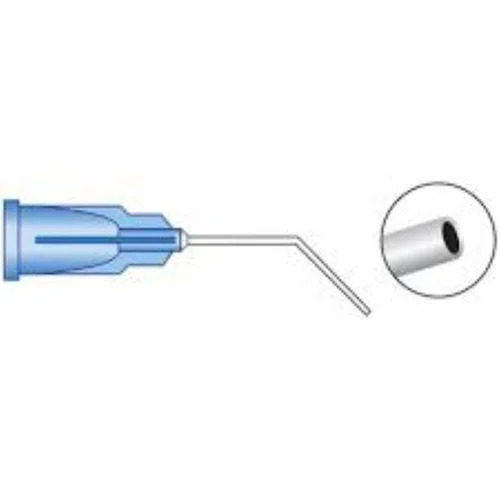
ANESTHESIA DISPOSABLE STERILE OPHTHALMIC RETROBULBAR/PERIBULBAR/SUB TENON CANNULA-19/21/23/25/27G
Price 20.0 INR/ Piece
ANESTHESIA DISPOSABLE STERILE OPHTHALMIC RETROBULBAR/PERIBULBAR/SUB TENON CANNULA-19/21/23/25/27G Specification
- Equipment Type
- Ophthalmic Anesthesia Cannula
- Material
- Medical grade stainless steel, flexible PVC
- Technology
- Precision extrusion and assembly
- Properties
- Disposable, Sterile, Flexible, Smooth Tip
- Grade
- Surgical Grade
- Driven Method
- Manual
- Dimension (L*W*H)
- Length: 25-38 mm (size dependent)
- Weight
- Approx. 1-3 g (size dependent)
- Usage
- Ophthalmic regional anesthesia (retrobulbar, peribulbar, sub-Tenon)
- Sterilized
- Yes
- Sterilization Mode
- Ethylene Oxide Gas (EO) sterilized
- Latex Content
- Latex free
- Shelf Life
- Approximately 5 years (unopened, sterile)
- Gauge Size Options
- 19G, 21G, 23G, 25G, 27G
- Connector
- Luer lock for secure syringe attachment
- Packaging
- Individually packed in sterile blister or pouch
- Intended Procedure
- Regional ocular anesthesia retrobulbar, peribulbar, sub-Tenon injection
- Tip Design
- Rounded, smooth, blunt end to minimize tissue trauma
ANESTHESIA DISPOSABLE STERILE OPHTHALMIC RETROBULBAR/PERIBULBAR/SUB TENON CANNULA-19/21/23/25/27G Trade Information
- Minimum Order Quantity
- 100 Pieces
- Delivery Time
- 2 Days
About ANESTHESIA DISPOSABLE STERILE OPHTHALMIC RETROBULBAR/PERIBULBAR/SUB TENON CANNULA-19/21/23/25/27G
Anesthesia Disposable Cannula-19g/21g/23g/25g/27g Disposable ophthalmic cannula used for administering local anesthesia.
Anesthesia disposable sterile ophthalmic cannulas are specialized, single-use medical instruments designed to safely deliver local anesthetic agents into the retrobulbar, peribulbar, or sub-Tenon's space around the eye for various ophthalmic surgeries. These techniques provide effective pain control (analgesia) and eye muscle paralysis (akinesia) with varying safety profiles.
| Anesthesia Type | Injection Site | Cannula/Needle Characteristics | Key Features |
|---|---|---|---|
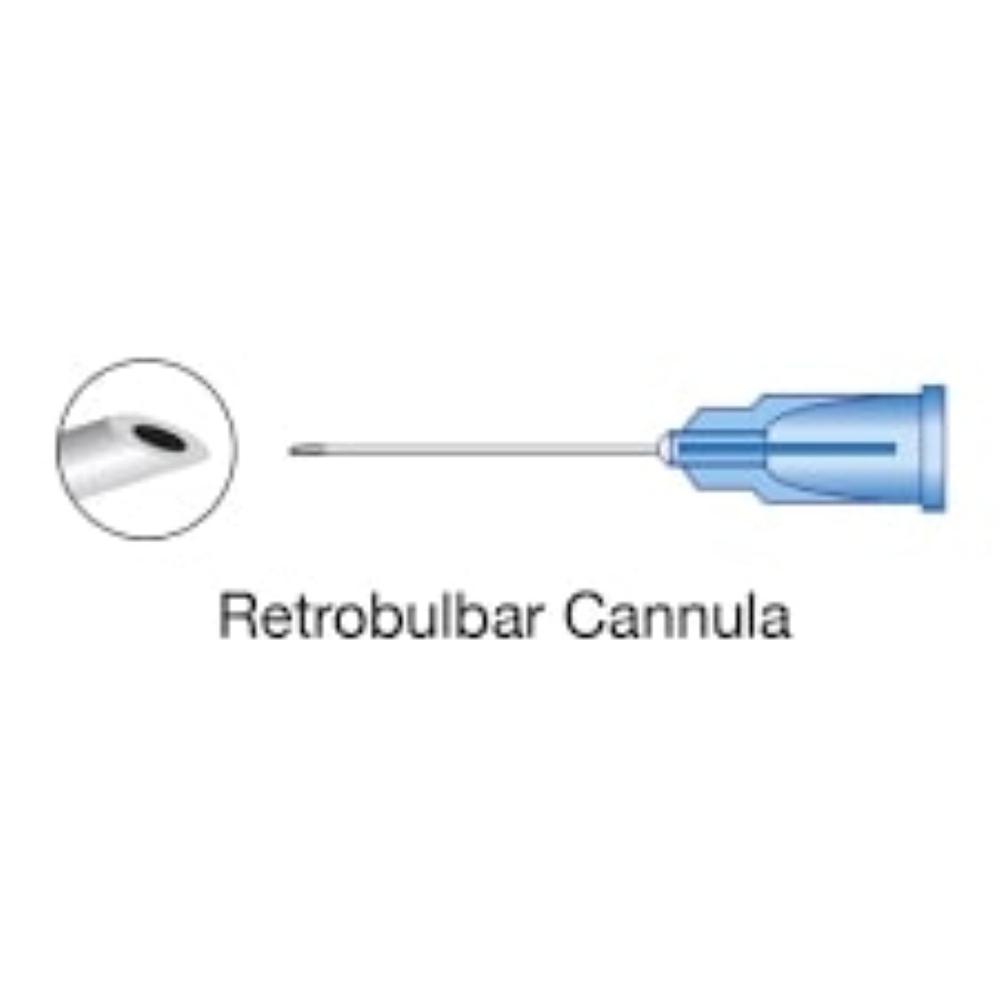
| Retrobulbar | Into the muscle cone behind the globe | Typically a sharp, 23- or 25-gauge needle, 30-38 mm in length | Provides faster, more complete akinesia but carries a higher risk of rare, serious complications |
| Peribulbar | Into the extraconal space, around the equator of the eyeball | Usually a sharp needle, shorter than 25 mm | Considered safer than retrobulbar but may require a larger volume of anesthetic and has a slower onset of action |
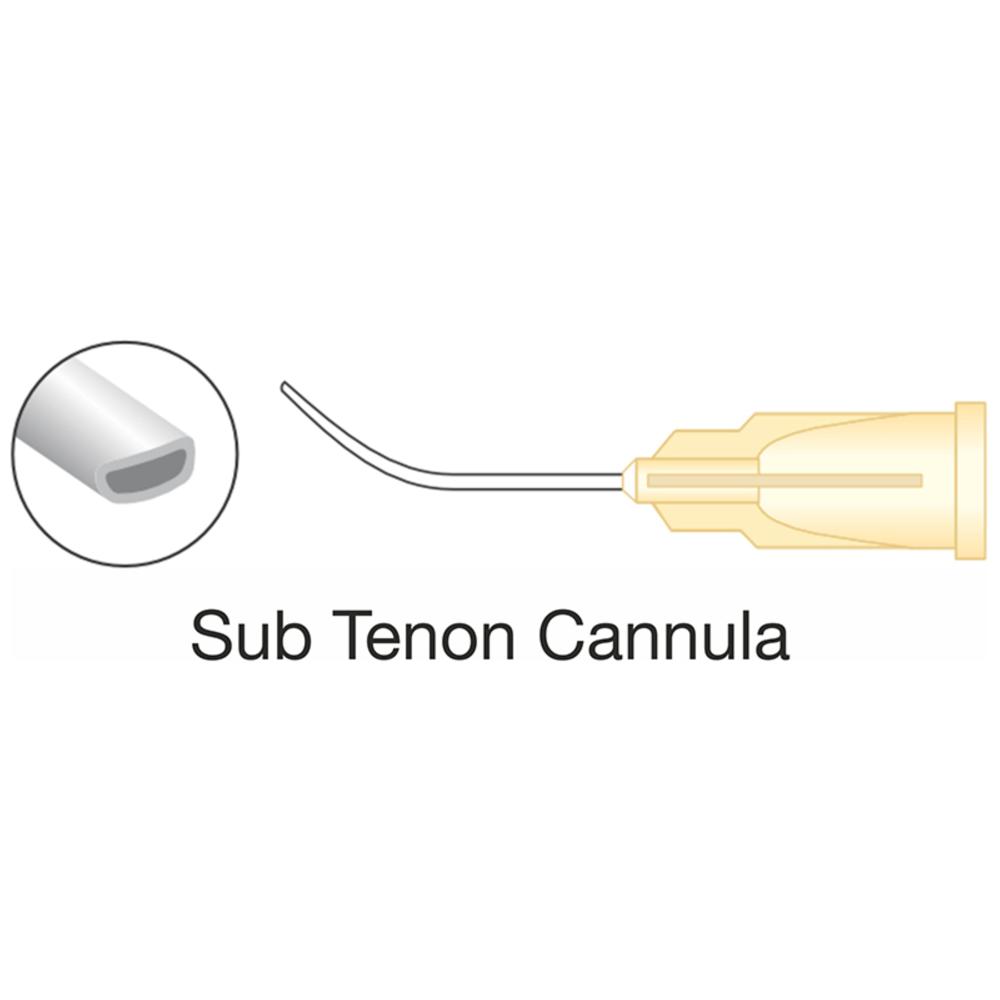
| Sub-Tenon's | Into the episcleral space (between the sclera and Tenon's capsule) | Blunt-tipped cannula, often curved or angled, with a spatulated or tri-port tip, e.g., 19G, 21G, or 23G with lengths around 25mm to 35mm | Offers a good balance of safety and efficacy, avoiding sharp needle-related risks; the blunt design minimizes tissue trauma |
Versatile Gauge Selection For All Needs
With options ranging from 19G to 27G, clinicians can select the most appropriate gauge for each type of ophthalmic regional anesthesia. The varied lengths and diameters facilitate flexibility and optimal performance during retrobulbar, peribulbar, and sub-Tenon procedures, catering to diverse anatomical and procedural requirements.
Precision Engineering for Patient Comfort
Every cannula is constructed using high-grade stainless steel and flexible PVC, complemented by a meticulously rounded blunt tip that helps minimize trauma to delicate ocular tissues. The Luer lock connector ensures secure and convenient syringe attachment, contributing to a smoother, safer injection process.
Sterility and Safety Assured
Each cannula is individually blister-packed and EO gas sterilized, ensuring maximum sterility right up to the point of use. They are latex-free, reducing the risk of allergic reactions, and are disposable, eliminating the need for reprocessing, and enhancing patient safety in clinical environments.
FAQ's of ANESTHESIA DISPOSABLE STERILE OPHTHALMIC RETROBULBAR/PERIBULBAR/SUB TENON CANNULA-19/21/23/25/27G:
Q: How do I select the appropriate gauge size for ophthalmic anesthesia procedures?
A: Choose the gauge size based on the specific anesthesia technique and patient anatomy. Thicker gauges (e.g., 19G and 21G) may be preferred for denser tissues or when larger anesthetic volumes are needed, while finer gauges (such as 25G and 27G) are typically used for delicate areas or to minimize tissue disruption.Q: What is the primary benefit of using a blunt, rounded tip on the cannula?
A: A blunt, rounded tip helps minimize trauma to ocular tissues during injection, reducing the risk of complications like hemorrhage or inadvertent penetration. This design promotes safer, more comfortable administration of anesthesia during retrobulbar, peribulbar, or sub-Tenon procedures.Q: When should this disposable cannula be used in clinical settings?
A: These cannulas are intended for single-use during ophthalmic procedures that require regional anesthesia, such as retrobulbar, peribulbar, or sub-Tenon blocks. They should be used by trained medical professionals immediately after removing from the sterile packaging.Q: Where is this cannula most commonly utilized?
A: The product is most commonly used in hospitals, specialty eye clinics, surgical centers, and other clinical environments where regional ocular anesthesia is administered for ophthalmic surgeries or procedures.Q: What is the sterilization process for these cannulas?
A: Each cannula is sterilized using Ethylene Oxide (EO) gas and then individually sealed in a sterile, tamper-proof blister pouch to maintain its sterility until needed for use.Q: How do I benefit from the individual sterile packaging?
A: Each cannula's individual sterile packaging ensures it remains uncontaminated until the point of use, significantly reducing infection risk and eliminating the need for on-site reprocessing or sterilization.Q: What materials are used in the cannula, and are they safe for patients with latex allergies?
A: The cannula is manufactured from medical-grade stainless steel and flexible PVC, and is entirely latex-free, making it safe for patients with latex sensitivities or allergies.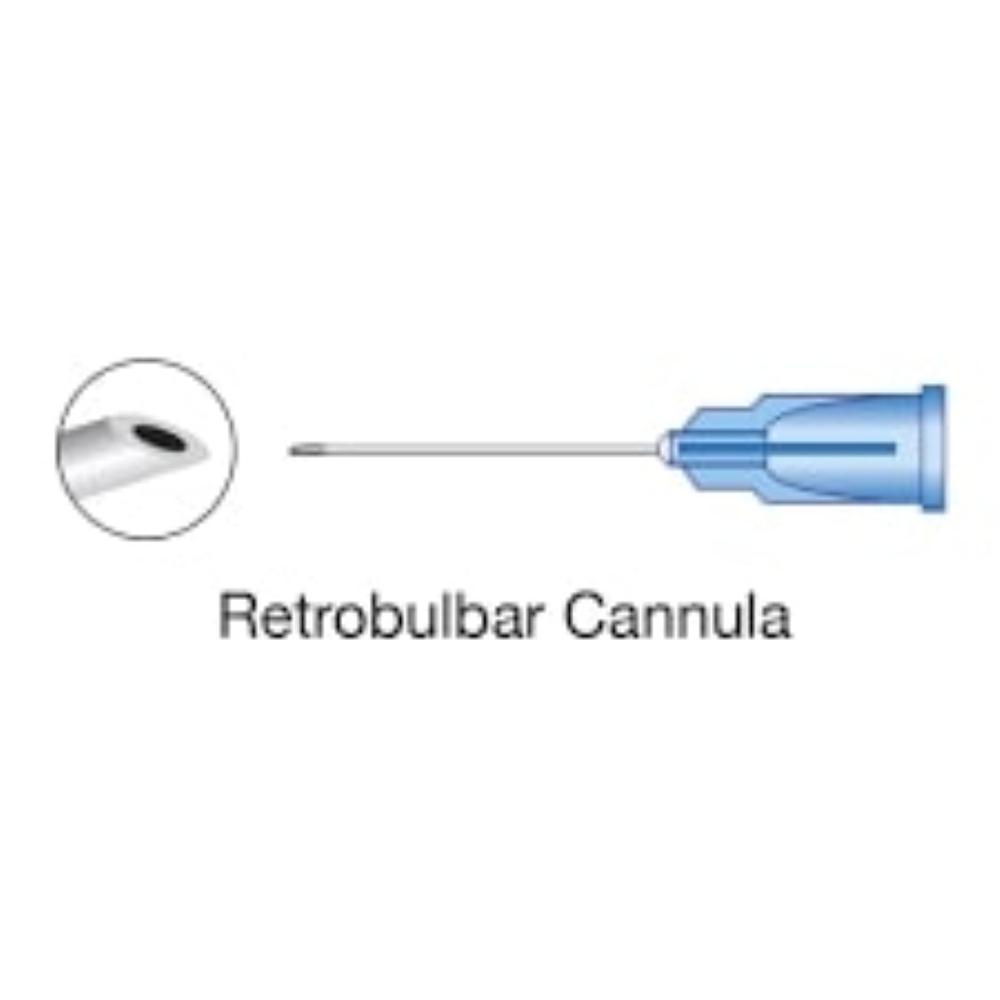

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Ophthalmic Cannula Category
Air Injection Disposable Cannula
Price 14.0 INR / Piece
Minimum Order Quantity : 200 Pieces
Color : Silver
Condition : New
Feature : Easy to use
Portable : Yes

 Send Inquiry
Send Inquiry